
In the 1950s, most inorganic chemistry textbooks defined transition elements as finishing at group 10 (nickel, palladium and platinum), therefore excluding group 11 (copper, silver and gold), and group 12 (zinc, cadmium and mercury). Which elements start to be counted as post-transition metals depends, in periodic table terms, on where the transition metals are taken to end. Elements 112–118 (copernicium, nihonium, flerovium, moscovium, livermorium, tennessine, and oganesson) may be post-transition metals insufficient quantities of them have been synthesized to allow sufficient investigation of their actual physical and chemical properties. If so, it would be a post-transition metal. Astatine, which is usually classified as a nonmetal or a metalloid, has been predicted to have a metallic crystalline structure. Other elements sometimes included are the group 11 metals copper, silver and gold (which are usually considered to be transition metals) the group 12 metals zinc, cadmium and mercury (which are otherwise considered to be transition metals) and aluminium, germanium, arsenic, selenium, antimony, tellurium, and polonium (of which germanium, arsenic, antimony, and tellurium are usually considered to be metalloids). Usually included in this category are the group 13–15 metals in periods 4–6: gallium, indium and thallium tin and lead and bismuth. This separation extends to other differences in physical and chemical behaviour between the light and heavier alkaline earth metals. The increased electronegativity of Be and Mg and the higher melting point of Be distances these light alkaline earth metals from their heavier congeners. Starting from the bottom left, and proceeding clockwise, the alkali metals are followed by the heavier alkaline earth metals the rare earths and actinides (Sc, Y and the lanthanides being here treated as rare earths) transition metals with intermediate electronegativity values and melting points the refractory metals the platinum group metals and the coinage metals leading and forming a part of the post-transition metals. 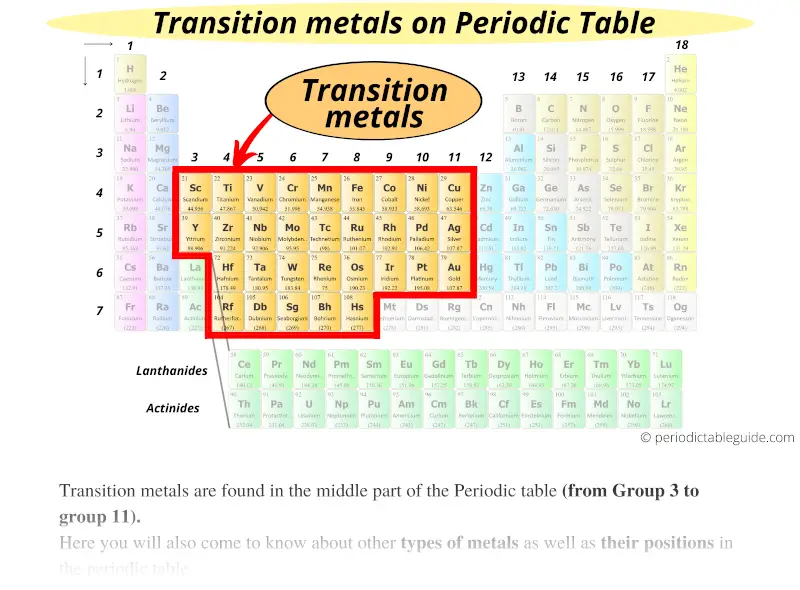
The geography of the plot broadly matches that of the periodic table. High electronegativity corresponds to increasing nonmetallic character low melting temperature corresponds to weaker cohesive forces between atoms and reduced mechanical strength. Elements categorised by some authors as post-transition metals are distinguished by their relatively high electronegativity values and relatively low melting points. Scatter plot of electronegativity values and melting points for metals (up to fermium, element 100) and some borderline elements (Ge, As, Sb, At). Alternate names for this group are B-subgroup metals, other metals, and p-block metals and at least thirteen other labels. The origin of the term is unclear: one early use was in 1940 in a chemistry text. The name is universally used, but not officially sanctioned by any organization such as the IUPAC. 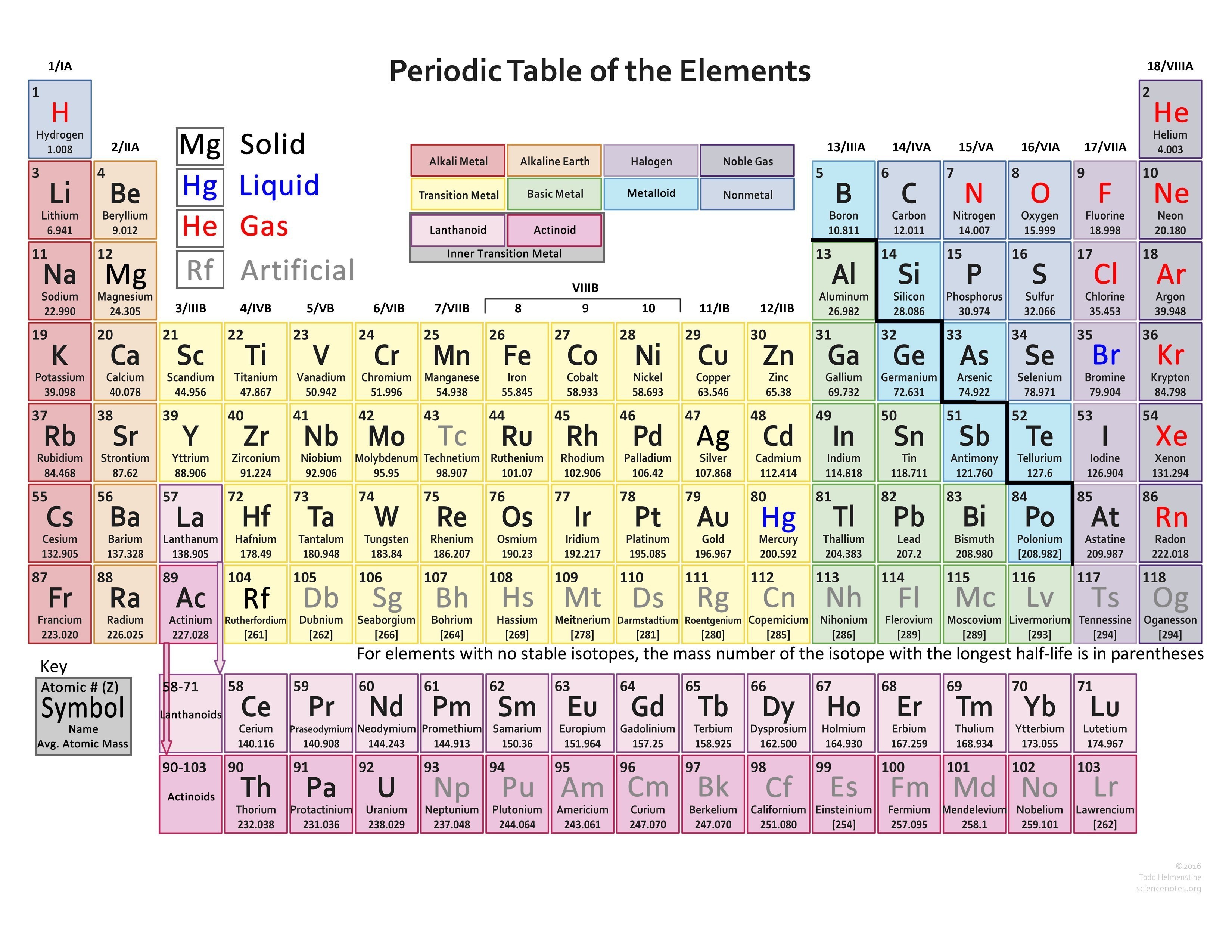
They can also form Zintl phases (half-metallic compounds formed between highly electropositive metals and moderately electronegative metals or metalloids). Chemically, they are characterised-to varying degrees-by covalent bonding tendencies, acid-base amphoterism and the formation of anionic species such as aluminates, stannates, and bismuthates (in the case of aluminium, tin, and bismuth, respectively). Being close to the metal-nonmetal border, their crystalline structures tend to show covalent or directional bonding effects, having generally greater complexity or fewer nearest neighbours than other metallic elements. Physically, post-transition metals are soft (or brittle), have poor mechanical strength, and have melting points lower than those of the transition metals. All proposals include gallium, indium, tin, thallium, lead, and bismuth. Depending on where these adjacent groups are judged to begin and end, there are at least five competing proposals for which elements to include: the three most common contain six, ten and thirteen elements, respectively (see image). Post-transition metals are a set of metallic elements in the periodic table located between the transition metals to their left, and the metalloids to their right. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
